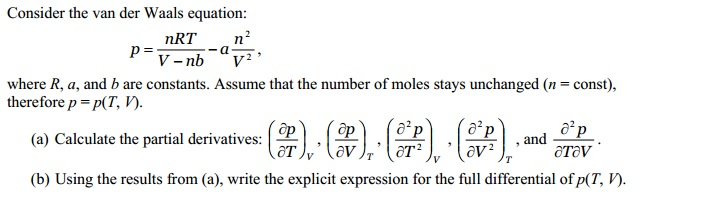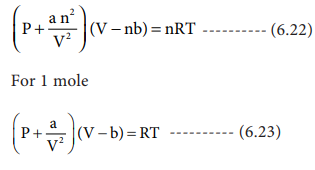= RT , at high pressure, the Van der Waals equation gets reduced to : Using Van der Waals equation, [ P + a/V^2 ](V - b) = RT , at high pressure, the Van der Waals equation gets reduced to :](https://dwes9vv9u0550.cloudfront.net/images/9876945/d6a3d03b-a449-4d51-986d-b05943344e70.jpg)
Using Van der Waals equation, [ P + a/V^2 ](V - b) = RT , at high pressure, the Van der Waals equation gets reduced to :

Using the van der Waals equation, calculate the pressure of 10.0mol NH(3) gas in a 10.0 L vessel at 27^(@)C. {:((P+n^(2)(a)/(V^(2)))(V -nb)=nRT,,a=4.2L^(2).atm//mol^(2),,b=0.037L//mol):}
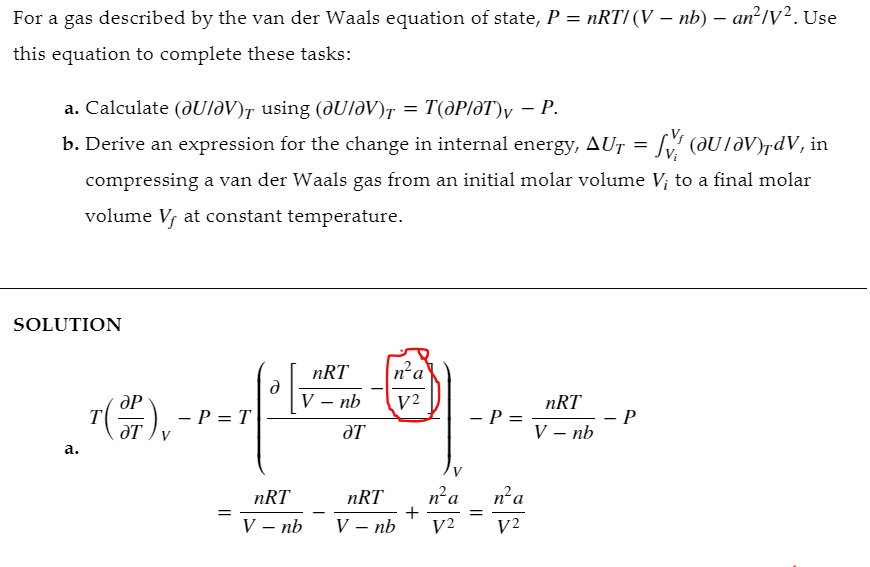
SOLVED: For a gas described by the van der Waals equation of state, P = nRTI (V nb) an?/V? . Use this equation to complete these tasks: Calculate (JUIdV)r using (JUIIV)T T(PIdT)v -

Using the van der Waals equation, calculate the pressure of 10.0mol NH(3) gas in a 10.0 L vessel at 27^(@)C. {:((P+n^(2)(a)/(V^(2)))(V -nb)=nRT,,a=4.2L^(2).atm//mol^(2),,b=0.037L//mol):}

SOLVED:Express the van der Waals equation of state as a virial expansion in powers of 1 / Vm and obtain expressions for B and C in terms of the parameters a and