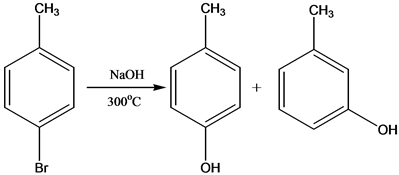Scheme 1. (i) and (iii) NaOH/p-toluenesulfonyl chloride, (ii) K 2 CO 3... | Download Scientific Diagram

White phosphorus on reaction with concentrated NaOH solution in an inert atmosphere of CO2 gives phosphine and compound (X) . (X) on acidification with HCl gives compound (Y) . The basicity of

Immobilization and characterization of Fe(0) catalyst on NaOH-treated coal fly ash for catalytic reduction of p-nitrophenol - ScienceDirect

Unexpected Complexity in the Products Arising from NaOH-, Heat-, Amine-, and Glycosylase-Induced Strand Cleavage at an Abasic Site in DNA | Chemical Research in Toxicology

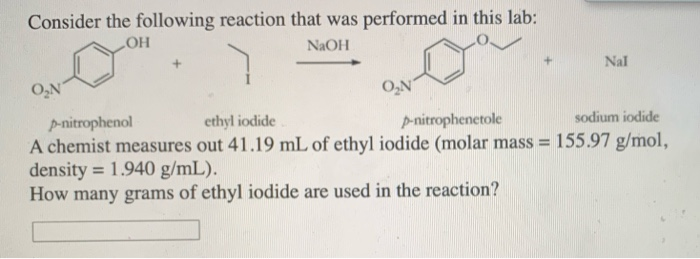
Predict the product of the reaction of p-methylbenzoic acid with the stated reagent. NaOH, then CH3I | Homework.Study.com

Cell Wash Solution I /Naoh-D 2L Roche Reagent Modular P/D Cobas C702 - China Cell Wash Solution I /Naoh-D and Roche Reagent
Relative percentages of phosphorus forms (a, OP, NaOH-P, HCl-P) to TP... | Download Scientific Diagram

Write the Product Formed When P-nitro Chlorobenzene is Heated with Aqueous Naoh at 443k Followed by Acidification? - Chemistry | Shaalaa.com
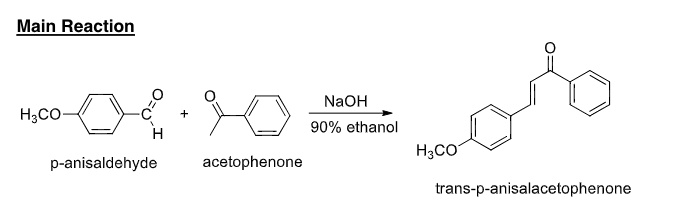
SOLVED: Main Reaction NaOH 90% ethanol HzCO acetophenone trans-p-anisalacetophenone HzCo p-anisaldehyde
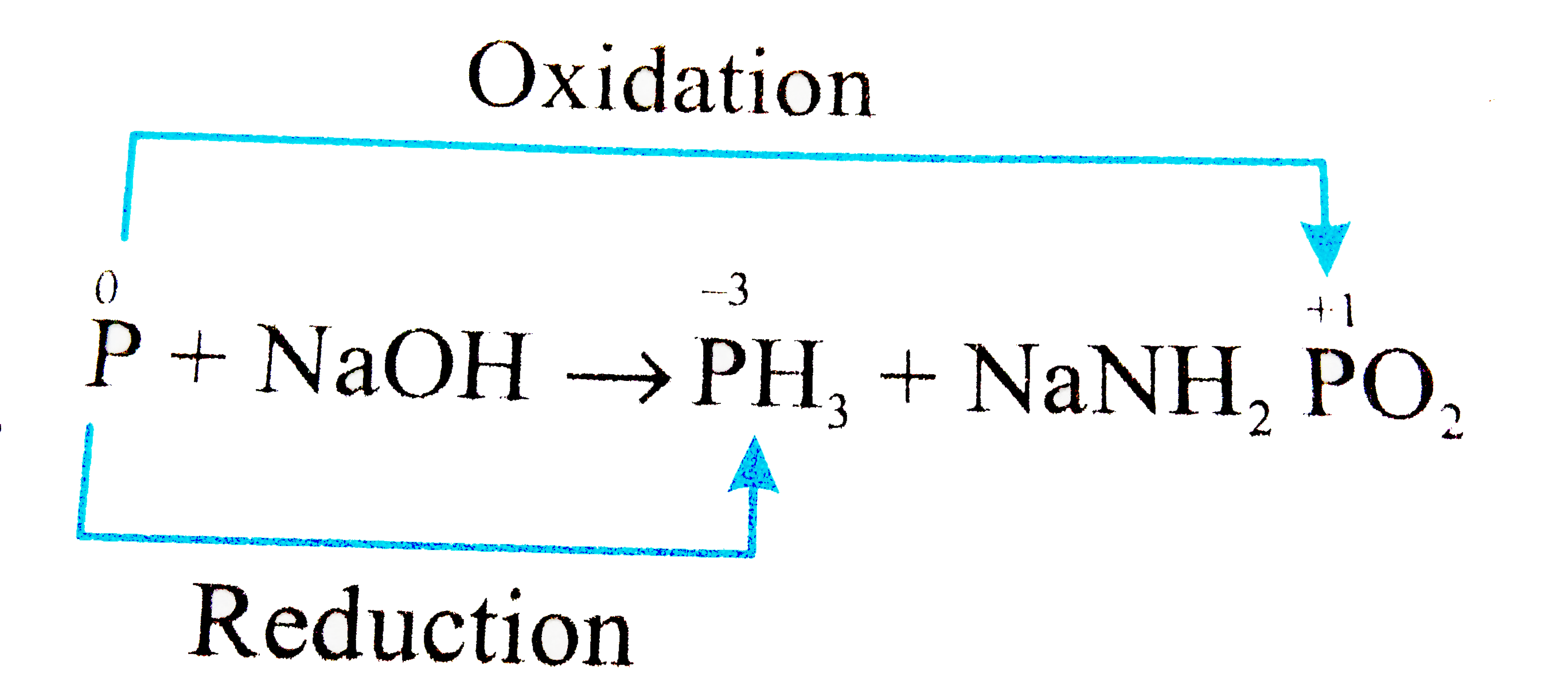
How should I balance this equation P + NaOH + H2O---------> PH3 + NaH2PO2 by ion electron method? - Quora
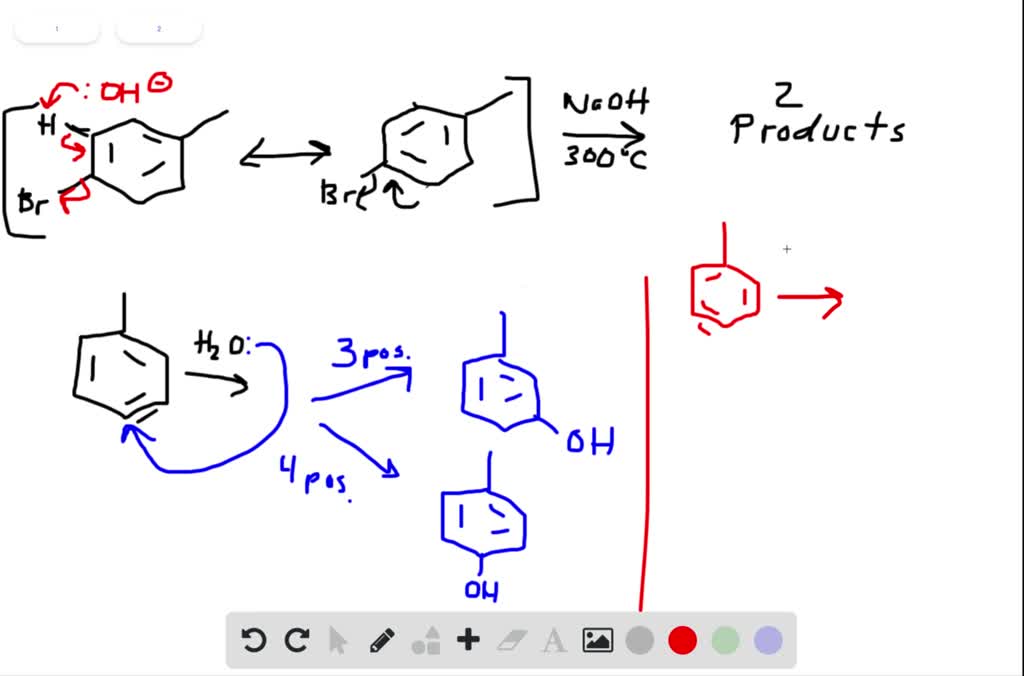
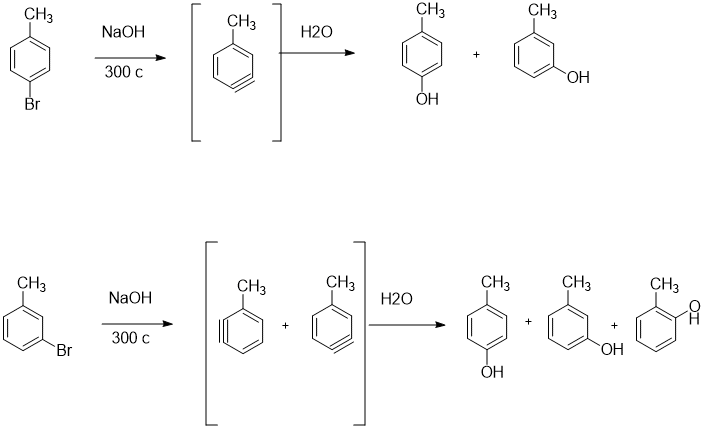
SOLVED:Treatment of p -bromotoluene with NaOH at 300^∘ C yields a mixture of two products, but treatment of m -bromotoluene with NaOH yields a mixture of three products. Explain.

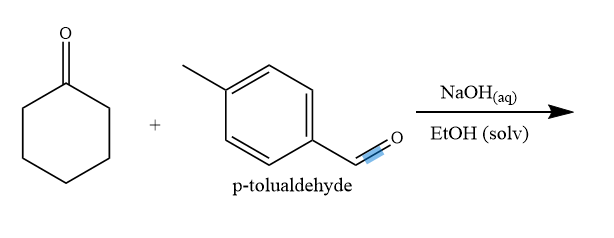



![ANSWERED] Consider the following three solutions of... - Physical Chemistry ANSWERED] Consider the following three solutions of... - Physical Chemistry](https://media.kunduz.com/media/sug-question/raw/59737718-1659709481.9039986.jpeg)