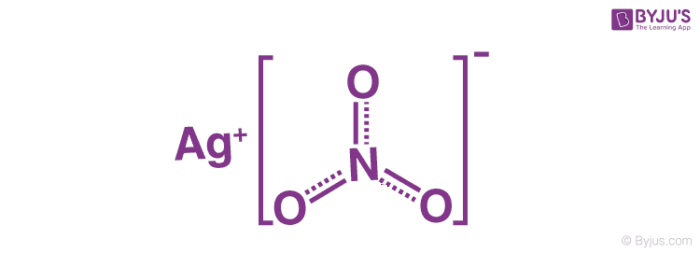
Scheme 3.2-The chemical synthesis of hepta-4,6-diyn-1-ol: i) AgNO3,... | Download Scientific Diagram

Thermo Scientific Chemicals Silver nitrate, ACS, 99.9+% (metals basis), Thermo Scientific Chemicals | Fisher Scientific
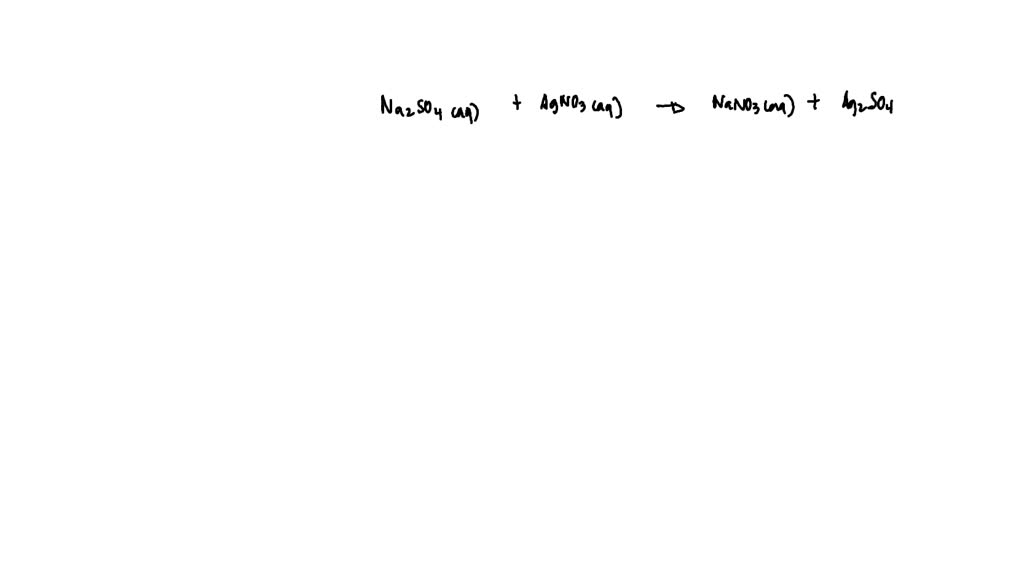
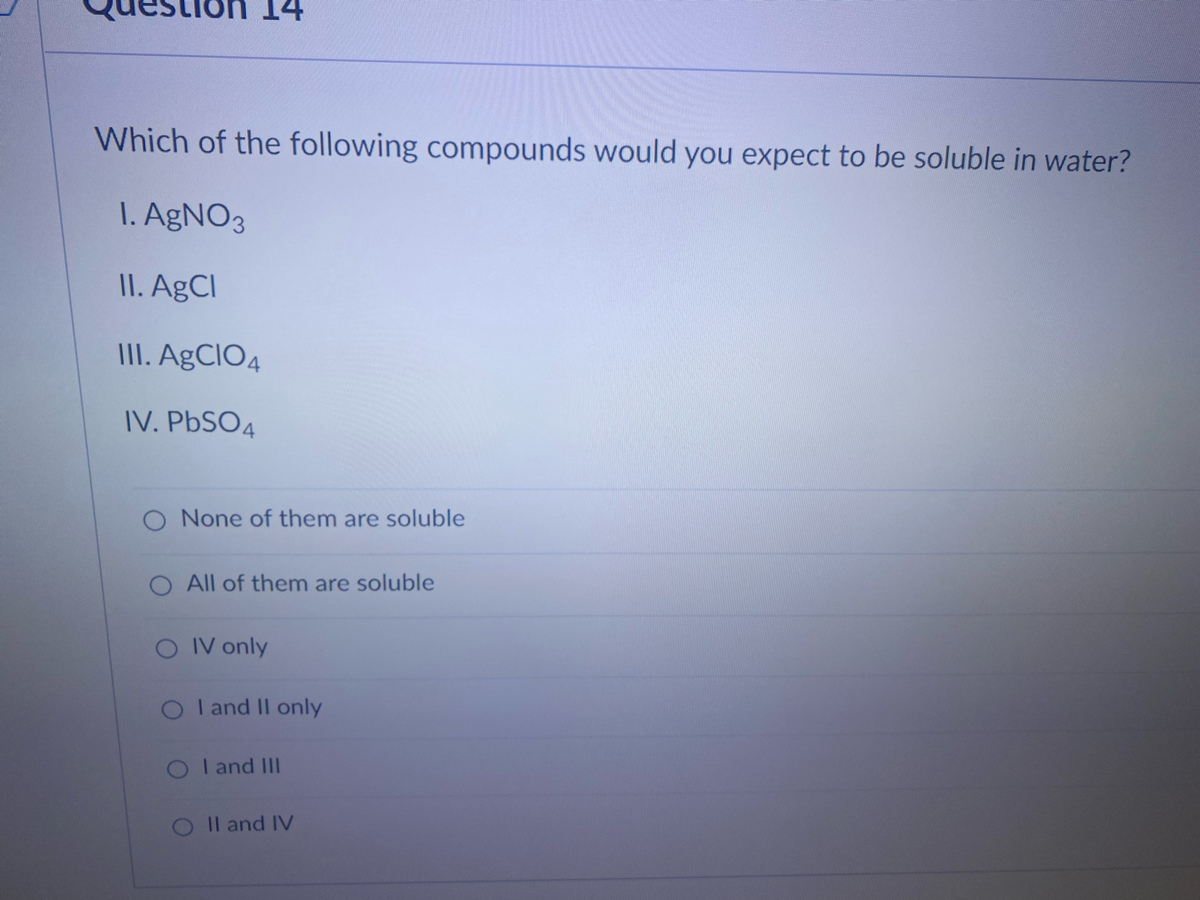
SOLVED: Which of the following solutions, when added to a solution of Na2SO4 would form a precipitate?I. AgNO3 (aq) II. KNO3 (aq) III. CaCl2 (aq)
![In the given reaction: CH3-CH2-COOH underset((ii) Br2,C Cl4//Delta)overset(( i) AgNO3)to [X]. [X] will be In the given reaction: CH3-CH2-COOH underset((ii) Br2,C Cl4//Delta)overset(( i) AgNO3)to [X]. [X] will be](https://d10lpgp6xz60nq.cloudfront.net/web-thumb/643045427_web.png)
In the given reaction: CH3-CH2-COOH underset((ii) Br2,C Cl4//Delta)overset(( i) AgNO3)to [X]. [X] will be

Explain the following :(i) When NaCl is added to AgNO3 solution, a white precipitate is formed.(ii) An aqueous solution of ammonium chloride is acidic in nature.
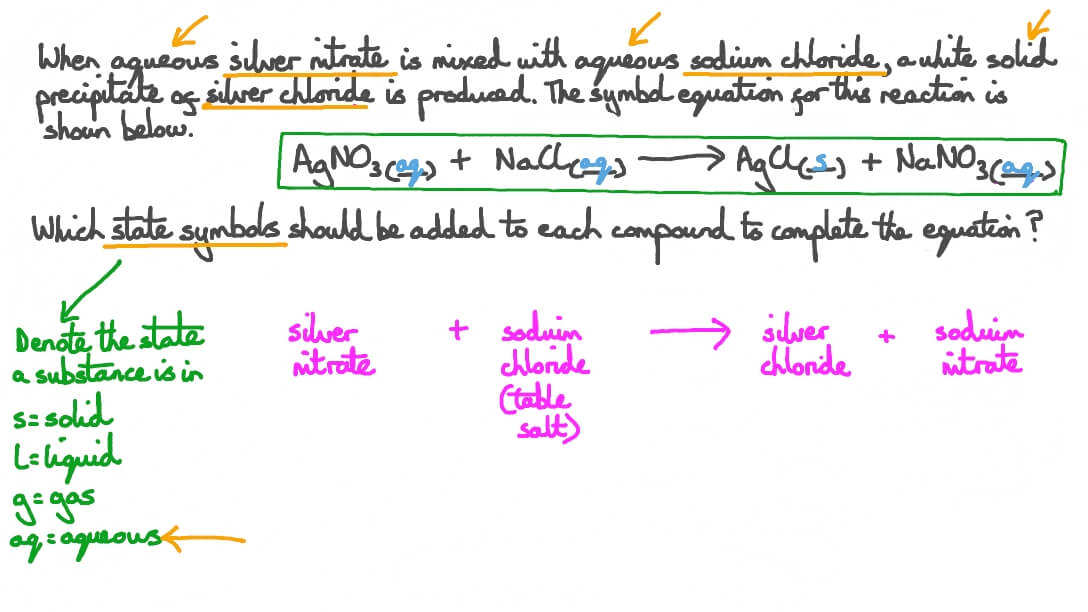
Question Video: Symbol Equation for the Reaction of the Precipitation of Agno3 with State Symbols | Nagwa










![In the given reaction CH3 - CH2 - COOH [ (ii)Br2/Δ ] (i)AgNO3 [ X ] , [ X ] will be : In the given reaction CH3 - CH2 - COOH [ (ii)Br2/Δ ] (i)AgNO3 [ X ] , [ X ] will be :](https://dwes9vv9u0550.cloudfront.net/images/10618672/ca1d5809-a712-4005-aea8-cf983d81c254.jpg)