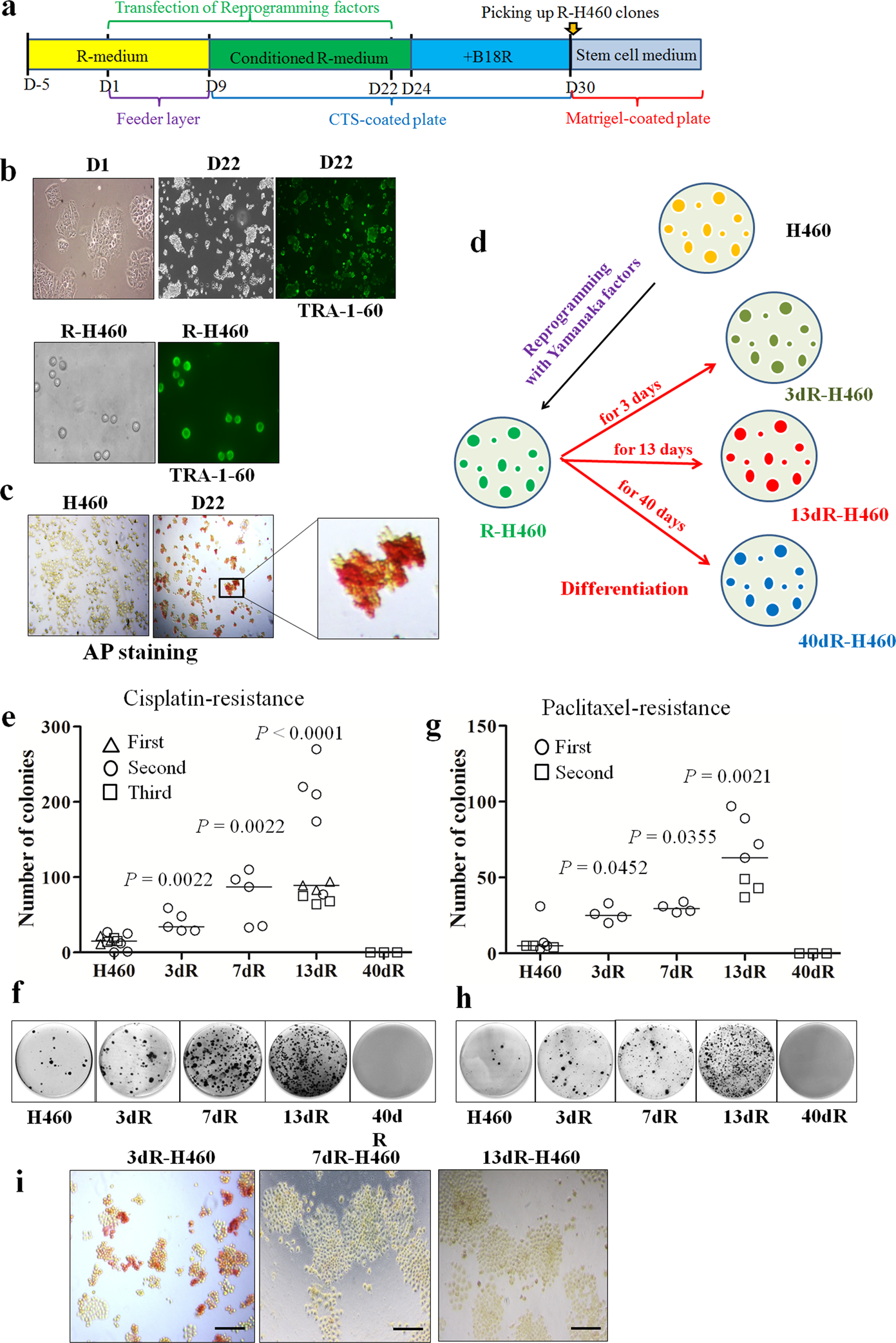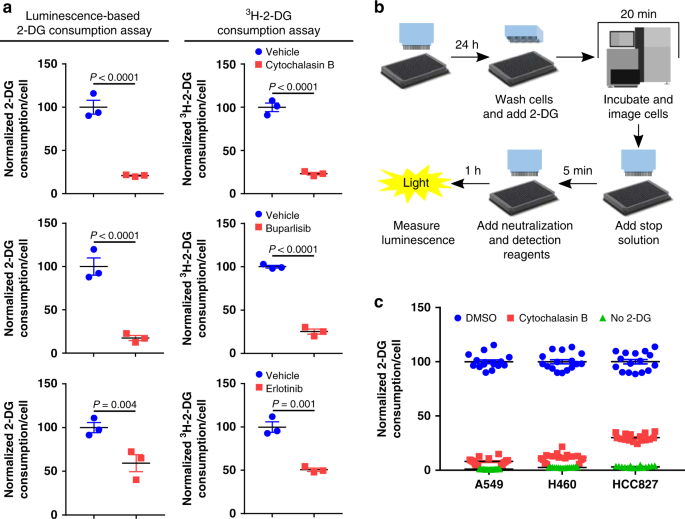
A high-throughput screen identifies that CDK7 activates glucose consumption in lung cancer cells | Nature Communications

Suppression of endothelial miR-22 mediates non-small cell lung cancer cell-induced angiogenesis: Molecular Therapy - Nucleic Acids
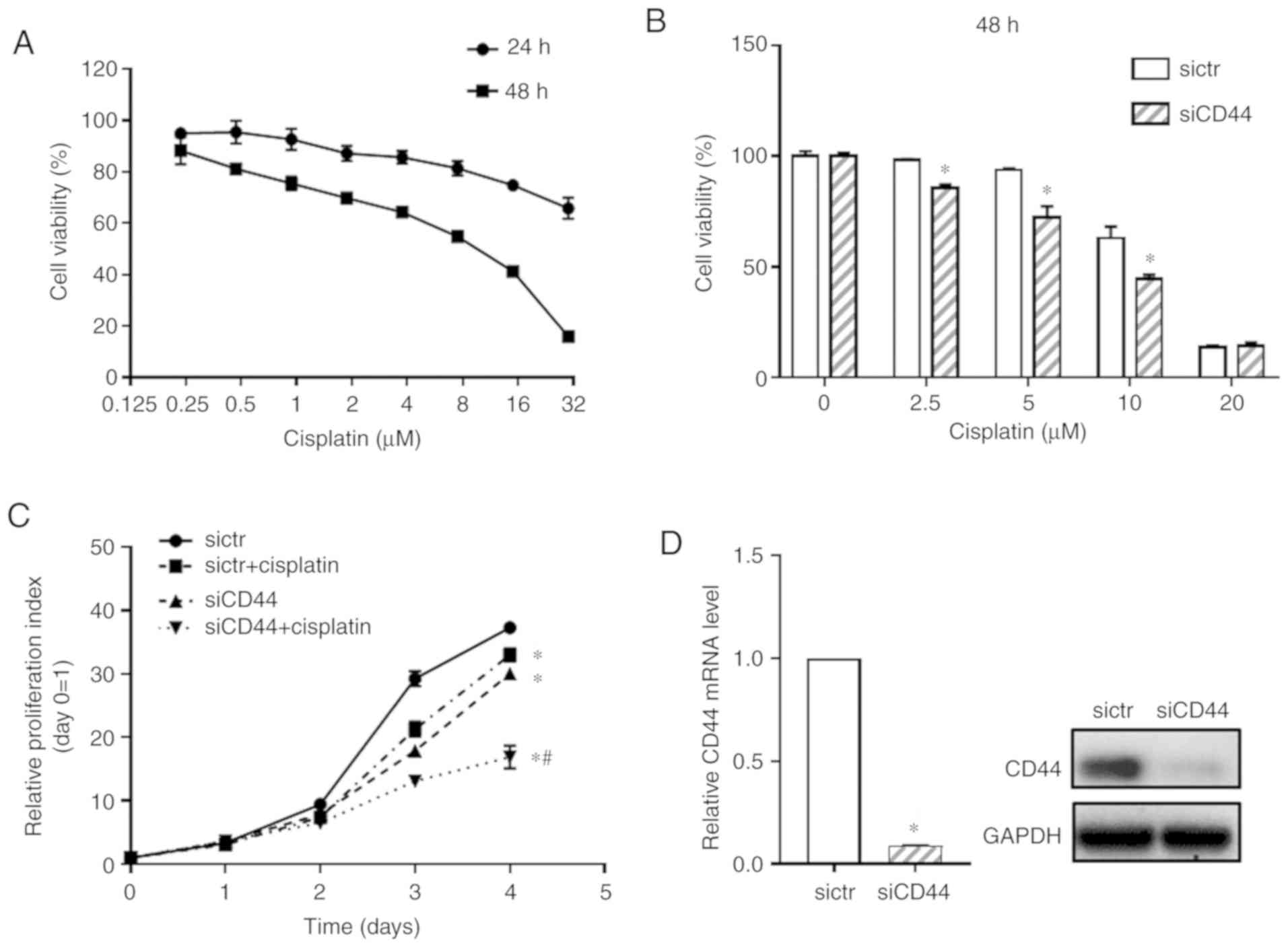
CD44 inhibition attenuates EGFR signaling and enhances cisplatin sensitivity in human EGFR wild‑type non‑small‑cell lung cancer cells

Enhancer remodeling promotes tumor-initiating activity in NRF2-activated non-small cell lung cancers | Nature Communications

Calculate the heat of combustion of ethene CH2 = CH2(g) + 3O2(g)→ 2CO2(g) + 2H2O(I) The bond energy data are given below C = C = 619 kJ mol^-1 C - H =
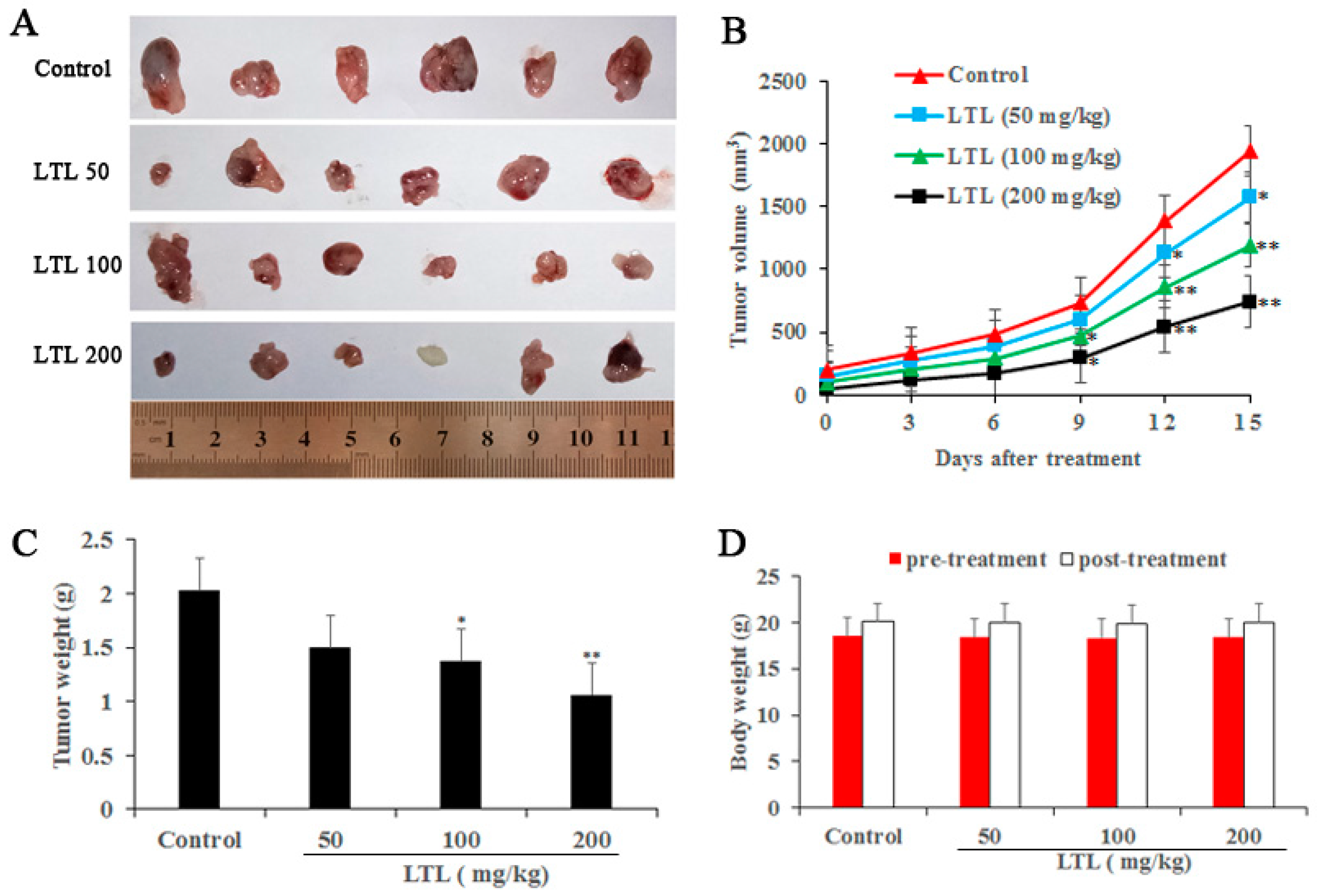
IJMS | Free Full-Text | Luteolin Inhibits Tumorigenesis and Induces Apoptosis of Non-Small Cell Lung Cancer Cells via Regulation of MicroRNA-34a-5p